 
Some chemical and physical properties of the halogens are summarized in Table G r o u p 17. The weaker electrostatic attraction of the electron being gained to the positive nucleus, makes it harder for the atom to gain an electron and it decreases in reactivity in group 7. Group 17: Physical Properties of the Halogens. Therefore, the attraction of the outer electron to the nucleus decreases as you go down group 7. There are more electrons shielding the positively charged nucleus from the outer electron. We are going to talk about the details of the mechanism below, but for now, let’s also mention that the reactivities of aryl halides increase, depending on the leaving group, in the following order: So, the more electronegative the halogen, the better leaving group it is in a nucleophilic aromatic substitution. 
They increase in electron shells so the atoms are larger as you go down the group. The atomic mass of the halogens increases.As you go down group 7, reactivity of the halogens decreases because: The negatively charged electron is attracted to the halogen atom due to the positive charge of the atom’s nucleus. 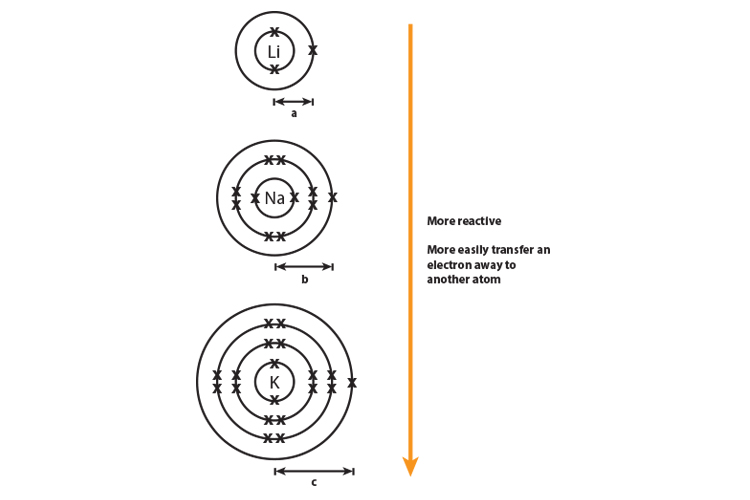
tivity trends can be engineered by deploying mixed halide. Halogens react to gain an electron so they become more stable. step in SMC reactions and the addition step in SNAr reactions have mechanistic similarities. The results provide another example of β-elimination reactions poised near the E2-E1cB mechanistic borderline.Group 7: Reactivity (GCSE Chemistry) Group 7: Reactivity Halogens – trend in reactivityĪs you look at the trend in reactivity of group 7, the reactivity of halogens decreases so fluorine is the most reactive halogen and astatine is the least reactive halogen. the most reactive halogen displaces all of the other halogens from solutions of their salts, and is itself displaced by none of the others. The approximately 50-fold greater reactivity of the 2- and 4-cyano substrates is attributed to the influence of the electron withdrawing cyano group in the deprotonation step. Although no common reactivity trend can be produced as it results from the. Commercially available arylating agents, high atom economy, and high regioselectivity. though, when the halogen is strongly, electron-withdrawing but unreactive as. Upon treatment with KHMDS, CO bond formation occurs between carbohydrate alcohols and a diverse range of fluorinated (hetero)aromatics to provide the targets in good to excellent yields. 1 However, in common usage, the term is often limited to a fragment that departs with a pair of electrons in heterolytic bond cleavage. Computational results indicate that deprotonation of the H-bonded complex is probably barrier free, and is accompanied by simultaneous loss of the leaving group (E2) for L = Cl, Br, and I, but with subsequent, rapid loss of the leaving group (E1cB-like) for the poorer leaving groups, CN and F. A general and practical route to carbohydratearyl ethers by nucleophilic aromatic substitution (SNAr) is reported. In chemistry, a leaving group is defined by the IUPAC as an atom or group of atoms that detaches from the main or residual part of a substrate during a reaction or elementary step of a reaction. 
The reactions are second-order in, the mechanism involving rate determining hydrogen-bond formation between piperidine and the substrate-piperidine addition intermediate followed by deprotonation of this intermediate. A different leaving group order is observed in the substitution reactions of ring-substituted N-methylpyridinium compounds with piperidine in methanol: 2-CN ≥ 4-CN > 2-F ∼ 2-Cl ∼ 2-Br ∼ 2-I. The "element effect" in nucleophilic aromatic substitution reactions (SNAr) is characterized by the leaving group order, L = F > NO2 > Cl ≈ Br > I, in activated aryl substrates. Here are some general pointers for recognising the substituent effects: The H atom is the standard and is regarded as having no effect. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
